It is well-established that genetic diversity has been declining in many dog breeds over the past two centuries, as a result of strong selection on a number of specific traits, combined in some cases with reductions in breed population sizes at certain points in history as well as popular sire effects. This is not only a concern for many dog breeds and landraces, but is a concern for many domesticated species around the world.
A Loss of Genetic Diversity
Such loss of genetic diversity has a major impact on animal welfare, as loss of genetic diversity can make it difficult or impossible to maintain closed breeding populations over the long term while also maintaining a peak level of health and wellness in individual animals. Despite such concern, limited research has been conducted to understand the impact of using genetic tools available to dog breeders today to guide breeding with an aim to preserve diversity in established dog breeds.
Research published in the scientific journal Evolutionary Applications, by a team of Embark scientists and engineers, led by myself, Dr. Aaron Sams, Principal Scientist at Embark, uses population genetic simulations to understand the difference between using sparsely distributed data (e.g. sparse microsatellite/genetic marker panels) and densely distributed data (e.g. whole genome genotype panels) to inform individual breeding decisions.
Research Conclusions
This work supports the conclusions that we described in an earlier article based on preliminary data from this study. In this blog post we underscored that if using genetic testing to guide breeding decisions, you cannot assess and preserve diversity in regions of the genome that you do not analyze.
In this study, we simulated breeding decisions based on genetic similarity between pairs of dogs using a number of different statistical values, short pedigrees, and two different genetic assays: a sparsely distributed 33 marker panel and a dense genotype panel.
The team’s results showed that using the smaller 33 marker panel to produce “diverse” offspring had serious limitations. With this panel, roughly 95 percent of the genome is not directly nearby (or “linked” to) the markers used to measure genetic diversity. In these unlinked regions genetic variation, at best, looked similar to what the team observed from random mating simulations. At worst, this smaller panel actually led to more loss of diversity than if no genetic information (including pedigrees) was used at all. However, as expected, this panel did lead to preservation of diversity in the ~5 percent of the genome linked to the 33 markers in the panel.
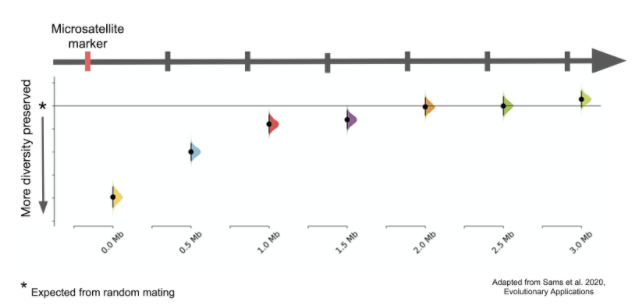
In contrast, using genome-wide genetic data led to more genetic variation preserved across the entire genome. This means that if genetic data is used alone, using a sparsely distributed genetic panel may do more harm than good.
If sparse microsatellite panels are not effective for retaining genome wide genetic diversity, why are they still used? Scientists still have reasons to use microsatellite panels, for example, for understanding genetic structure in populations where higher quality genetic information (e.g. from dense genotyping or resequencing) are not available. Microsatellites used in conjunction with pedigrees can also help ensure pedigree relationships are accurate. As a scientific tool, they have their place, but scientifically there is no evidence to support the routine use of these panels as a tool for breeders and breed clubs looking to preserve genetic diversity within their respective breeds.














